AI Theatre Session: Interoperability
AI-SC 9 - Post-market surveillance and quality assurance of AI tools
2 min
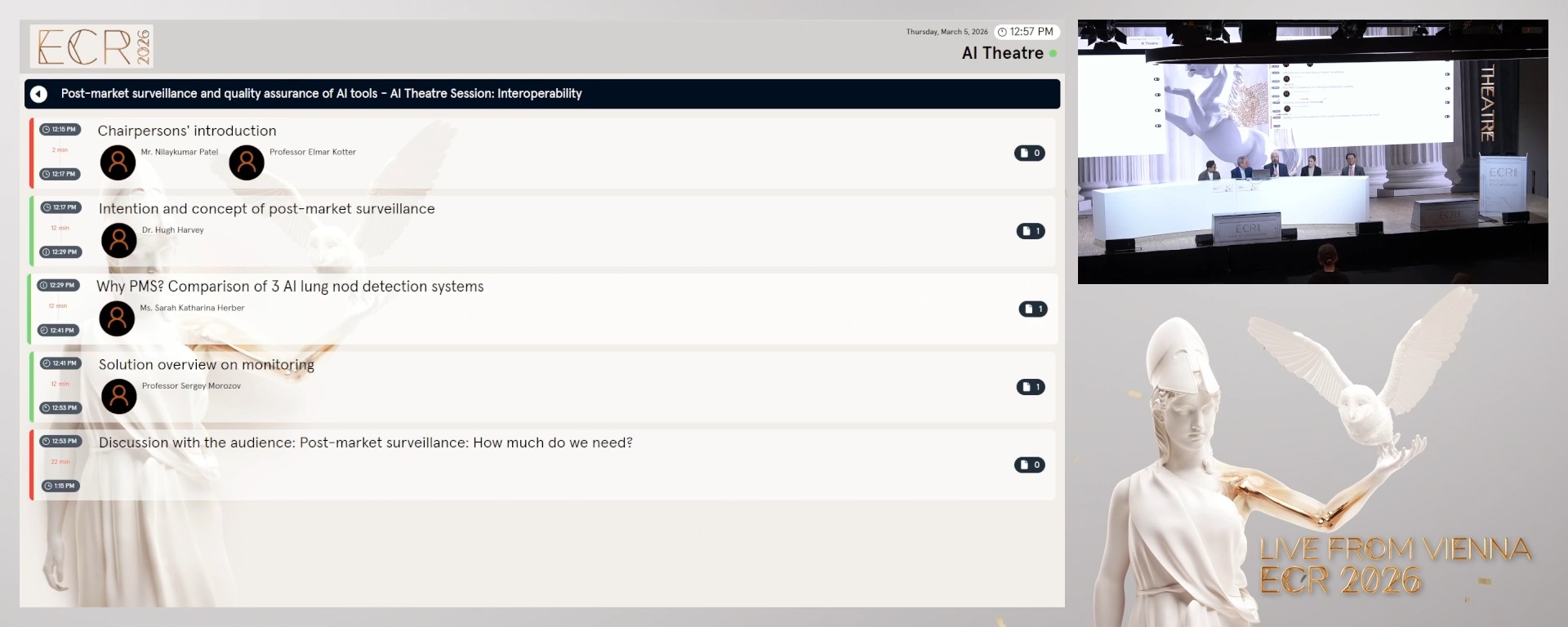
Chairpersons' introduction
Elmar Kotter, Freiburg Im Breisgau / Germany
Nilaykumar Patel, Vienna / Austria
Nilaykumar Patel, Vienna / Austria
12 min
Intention and concept of post-market surveillance
Hugh Harvey, Banstead / United Kingdom
- To learn what the medical device regulations in the EU mandate in regard to manufacturer responsibilities for PMS.
- To appreciate the burden placed on hospitals to assist manufacturers in conducting PMS effectively.
- To understand practical systems for managing AI post market surveillance.
12 min
Why PMS? Comparison of 3 AI lung nod detection systems
Sarah Katharina Herber, Mainz / Germany
- To learn how AI-based tools support lung nodule reporting, with particular strengths in detection and longitudinal follow-up.
- To appreciate the limitations of histopathological assessment for pulmonary nodules and how AI can complement, but not replace, current diagnostic pathways.
- To understand the clinical implications and future potential of AI-supported lung reporting, including workflow integration, patient management, and potential outcome improvement.
12 min
Solution overview on monitoring
Sergey Morozov, Brussels / Belgium
- To learn the fundamentals and techniques of continuous AI monitoring.
- To appreciate the variability and utility of objective and subjective metrics.
- To understand the practical approach to clinical AI monitoring.
22 min
Discussion with the audience: Post-market surveillance: How much do we need?